Word Equation For Potassium With Water
aseshop
Aug 29, 2025 · 6 min read
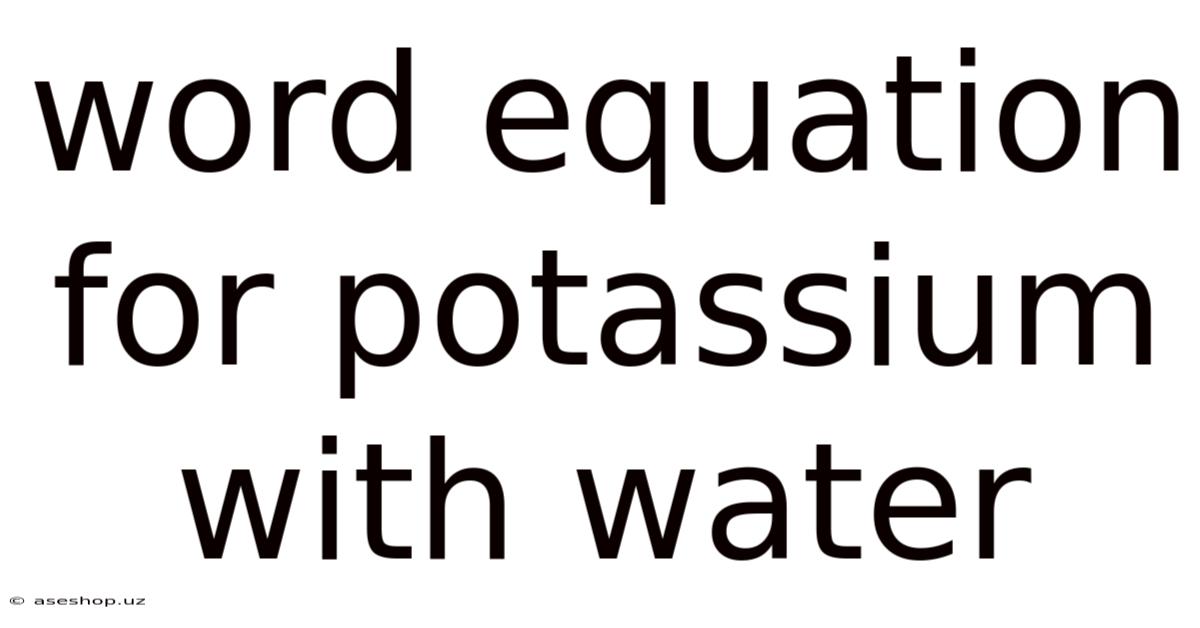
Table of Contents
The Energetic Reaction: Understanding the Word Equation and Beyond for Potassium and Water
Potassium (K) and water (H₂O) react vigorously, a reaction familiar to many chemistry students. This article delves deep into understanding this reaction, going beyond a simple word equation to explore the underlying chemistry, safety precautions, and practical applications. We'll examine the balanced chemical equation, explore the energetics involved, and discuss the fascinating implications of this seemingly simple interaction. Learning about this reaction provides a strong foundation for understanding more complex chemical processes.
Introduction: The Dramatic Reaction of Potassium and Water
The reaction between potassium and water is a classic example of a highly exothermic reaction, meaning it releases a significant amount of heat. This reaction is characterized by the rapid evolution of hydrogen gas (H₂) and the formation of potassium hydroxide (KOH), a strong alkali. The word equation simply summarizes this:
Potassium + Water → Potassium Hydroxide + Hydrogen
While this is a good starting point, a complete understanding requires a much deeper dive into the chemical processes at play. This article will guide you through the nuances of this reaction, providing a comprehensive picture of its chemical behavior and practical significance.
The Balanced Chemical Equation: More Than Just Words
The word equation provides a concise summary, but it lacks the precision of a balanced chemical equation. The balanced equation provides crucial information about the stoichiometry of the reaction, indicating the precise ratios of reactants and products. The balanced chemical equation for the reaction between potassium and water is:
2K(s) + 2H₂O(l) → 2KOH(aq) + H₂(g)
This equation tells us that two moles of solid potassium react with two moles of liquid water to produce two moles of aqueous potassium hydroxide and one mole of hydrogen gas. The “(s)” denotes solid, “(l)” denotes liquid, “(aq)” denotes aqueous (dissolved in water), and “(g)” denotes gas. Understanding this balanced equation is fundamental to predicting the quantities of reactants and products involved in the reaction.
A Step-by-Step Look at the Reaction Mechanism
The reaction between potassium and water isn't a simple one-step process. It involves several steps that contribute to the overall exothermicity and rapid nature of the reaction. Let's break down the mechanism:
-
Initial Contact: When potassium comes into contact with water, the highly reactive potassium atom readily donates its single valence electron to a water molecule. This electron transfer is the driving force behind the entire reaction.
-
Formation of Potassium Ion and Hydroxide Ion: The potassium atom loses its electron, forming a positively charged potassium ion (K⁺). The water molecule, having gained an electron, forms a hydroxide ion (OH⁻). This process is known as ionization.
-
Hydrogen Gas Evolution: The newly formed hydroxide ions are unstable and readily react with other water molecules, eventually leading to the formation of hydrogen gas (H₂). This is a complex process involving the transfer of protons (H⁺) and the breaking and formation of covalent bonds.
-
Dissolution of Potassium Hydroxide: The potassium hydroxide (KOH) produced is highly soluble in water, forming an aqueous solution. This solution is strongly alkaline, exhibiting a high pH.
-
Heat Release: The overall reaction is highly exothermic due to the strong bonds formed in potassium hydroxide and the liberation of hydrogen gas. The energy released is sufficient to ignite the hydrogen gas, often resulting in a visible flame.
The Energetics of the Reaction: Why is it so Exothermic?
The high exothermicity of the potassium-water reaction stems from several factors:
-
Low Ionization Energy of Potassium: Potassium has a relatively low ionization energy, meaning it requires relatively little energy to remove its valence electron. This facilitates the electron transfer to water, initiating the reaction.
-
Strong Bond Formation in KOH: The formation of potassium hydroxide involves the formation of strong ionic bonds between the K⁺ and OH⁻ ions. The energy released during bond formation is a major contributor to the exothermic nature of the reaction.
-
Bond Breaking in Water: While bond breaking requires energy input, the energy released during the formation of new bonds in KOH and H₂ far exceeds this input, resulting in a net release of energy.
-
Hydrogen Bond Disruption: The release of hydrogen gas disrupts the hydrogen bonding network in water, further contributing to the energy release.
Safety Precautions: Handling Potassium Requires Caution
Potassium is a highly reactive alkali metal. Direct contact with water can lead to a violent reaction with the potential for burns, fire, and explosion. Therefore, extreme caution must be exercised when handling potassium:
-
Always perform the reaction under a fume hood: The hydrogen gas produced is flammable, and the reaction generates heat and splashing.
-
Use small quantities of potassium: A small piece of potassium (a few milligrams) is sufficient to demonstrate the reaction.
-
Wear appropriate safety equipment: This includes safety goggles, gloves, and a lab coat.
-
Have a fire extinguisher readily available: Be prepared to extinguish any fire that might occur.
-
Never handle potassium with bare hands: Potassium reacts with moisture on the skin, causing burns.
Practical Applications: Beyond the Laboratory
While the reaction's primary use is in demonstrating chemical principles in the laboratory, the underlying chemistry has important applications:
-
Production of Potassium Hydroxide: Potassium hydroxide is a vital chemical used in various industrial processes, including the production of soaps, detergents, and fertilizers. While not the primary industrial method, the reaction demonstrates the fundamental chemical principle involved in KOH production.
-
Hydrogen Production (Theoretically): The reaction produces hydrogen gas. While not a cost-effective method for large-scale hydrogen production, it illustrates the potential of alkali metal reactions as a hydrogen source.
-
Research and Education: This reaction is widely used in educational settings to demonstrate the reactivity of alkali metals and the principles of exothermic reactions. It provides a captivating visual representation of fundamental chemical concepts.
Frequently Asked Questions (FAQs)
Q1: Why is the reaction between potassium and water more vigorous than the reaction between sodium and water?
A1: Potassium is more reactive than sodium because its valence electron is further from the nucleus and therefore more easily lost. This leads to a faster and more energetic reaction.
Q2: What are the products of the reaction?
A2: The products are potassium hydroxide (KOH) and hydrogen gas (H₂).
Q3: Is the reaction reversible?
A3: No, the reaction is essentially irreversible under normal conditions. The hydrogen gas escapes, and the potassium hydroxide remains dissolved in water.
Q4: What happens if a larger amount of potassium is used?
A4: Using a larger amount of potassium will result in a more vigorous reaction, potentially leading to a larger explosion and increased safety risks.
Q5: Can this reaction be used to generate electricity?
A5: The high energy released could theoretically be harnessed to generate electricity, although practical applications are limited due to the safety concerns and the need for controlled conditions.
Conclusion: A Powerful Demonstration of Chemical Principles
The reaction between potassium and water, while seemingly simple, offers a rich tapestry of chemical principles. From the balanced chemical equation to the detailed reaction mechanism and the energetic considerations, this reaction provides a powerful illustration of fundamental chemical concepts. Understanding this reaction enhances our knowledge of redox reactions, exothermic processes, and the reactivity of alkali metals. Always remember to prioritize safety when conducting this reaction or any experiment involving reactive chemicals. Through careful observation and a thorough understanding of the chemistry involved, we can unlock the fascinating insights hidden within this seemingly simple, yet dramatic, chemical interaction.
Latest Posts
Latest Posts
-
Two Vast And Trunkless Legs Of Stone Analysis
Aug 29, 2025
-
Lord Byron When We Two Parted
Aug 29, 2025
-
Explain How Amylase Breaks Down Starch
Aug 29, 2025
-
Difference Between Anaerobic Respiration And Aerobic Respiration
Aug 29, 2025
-
Act 3 Scene 2 Of Macbeth
Aug 29, 2025
Related Post
Thank you for visiting our website which covers about Word Equation For Potassium With Water . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.